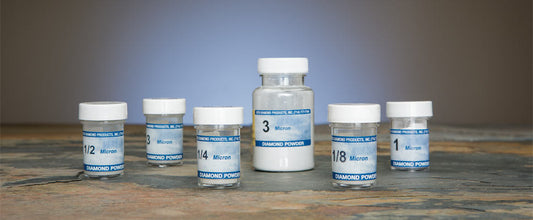
Collection: Metallographic Polishing
Request a complimentary sample — specify your material, equipment, and requirements. Ships within 2 business days.
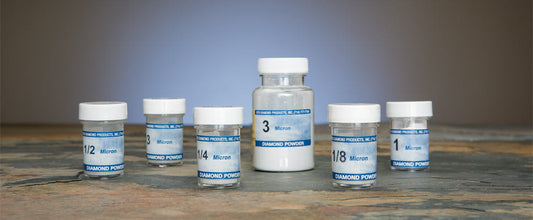
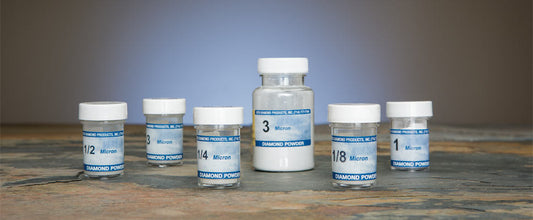
Beta Diamond Products supplies the complete range of polishing consumables for all stages of metallographic specimen preparation — from coarse diamond polishing through final oxide polishing for EBSD and high-resolution SEM analysis. Our polishing products include diamond suspensions, diamond compounds, colloidal silica polishing suspension, alumina polishing powders, and polishing cloths in all nap types for all material classes.
Metallographic Polishing Stages: What Each Step Requires
| Stage | Typical Abrasive | Cloth Type | Purpose |
|---|---|---|---|
| Coarse diamond polish | 9μm diamond suspension or compound | Hard woven cloth | Remove final grinding scratches; establish flat, uniform surface |
| Intermediate diamond polish | 3μm diamond suspension or compound | Medium nap cloth | Remove 9μm scratches; refine surface finish |
| Fine diamond polish | 1μm diamond suspension or compound | Low nap or silk cloth | Near-final polish; near-mirror surface on most metals |
| Final diamond polish | 0.25μm diamond suspension or compound | Low nap or silk cloth | Mirror-quality surface for optical metallography |
| Oxide polish (when required) | 0.06μm colloidal silica or 0.05μm alumina | Chemomechanical cloth | Remove residual deformation layer for EBSD, TEM, atom probe |
Colloidal Silica vs. Alumina for Final Oxide Polishing
Colloidal silica (OPS, 0.04μm) is the preferred final polishing medium for most FCC and HCP metals — titanium alloys, aluminum alloys, austenitic stainless steels, nickel superalloys, copper, and brass. The chemical-mechanical action of colloidal silica on these materials produces a deformation-free surface that gives clear Kikuchi patterns in EBSD without the artifacts that remain after pure mechanical polishing.
Alumina (Al₂O₃, 0.05–0.3μm) is preferred for BCC metals and alloys — ferritic steels, ferritic stainless steels, molybdenum, tungsten — where colloidal silica can cause preferential attack of grain boundaries or second phases. Alumina is also preferred for optical glass, ceramics, and substrates where surface chemistry must be tightly controlled.
Tell us your material, analysis technique, and polisher model and we'll specify the complete sequence. Request a free sample →
Frequently Asked Questions
What is colloidal silica used for in metallographic sample preparation?
Colloidal silica (typically 0.04–0.06μm particle size, also called OPS — Oxide Polishing Suspension) is used as the final polishing step for applications requiring a deformation-free surface. This includes EBSD (electron backscatter diffraction) analysis, orientation imaging microscopy, TEM cross-section preparation, and any application where the residual mechanical deformation layer introduced by diamond polishing would interfere with analysis. Colloidal silica acts by a combination of mechanical abrasion (from the fine SiO₂ particles) and mild chemical etching of the metal surface, producing a surface that is simultaneously polished and lightly etched at the atomic scale.
When should I use colloidal silica vs. alumina for final polishing?
Colloidal silica is preferred for FCC and HCP metals: titanium alloys, aluminum alloys, austenitic stainless steels, nickel superalloys, copper. Alumina (0.05–0.3μm) is preferred for BCC metals (ferritic steels, ferritic stainless steels, molybdenum) where colloidal silica can cause preferential grain boundary attack, and for optical glass and ceramics where surface chemistry must be controlled.
-
No reviews